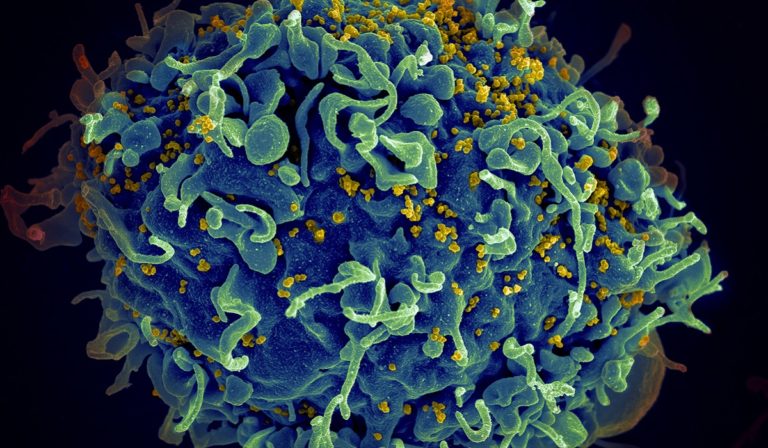
New twice-yearly shot to prevent HIV achieves 100% success rate in late-stage trial
In this double-blind, randomized study of 5,300 cisgender women in South Africa and Uganda, 2,134 got the injection and the others took one of two types of daily PrEP pills. The trial from California-based company Gilead Sciences began on August 2021 and, so far, not a single woman who received the injections has contracted HIV. The participants who received either of the oral PrEP options, Truvada and Descovy, had infection rates of about 2% — consistent with the infection rates of oral PrEP in other clinical trials.
New twice-yearly shot to prevent HIV achieves 100% success rate in late-stage trial Read more